NPRA is proud to announce the launch of our new website which coincides with our mission to safeguard the nation's health through scientific excellence in the regulatory control of medicinal products and cosmetics.
The website also boasts a clean design and intuitive with improved menu functionality that directs you to the information most relevant to you. It is also fully responsive with mobile devices, making it easy to navigate on a wide range of web browsers and portable devices.
Check out our improvement on the website :
- 3 Main division on the website which is Consumers, Health Professionals & Industry

2. Infographic display on consumers & health professionals main page

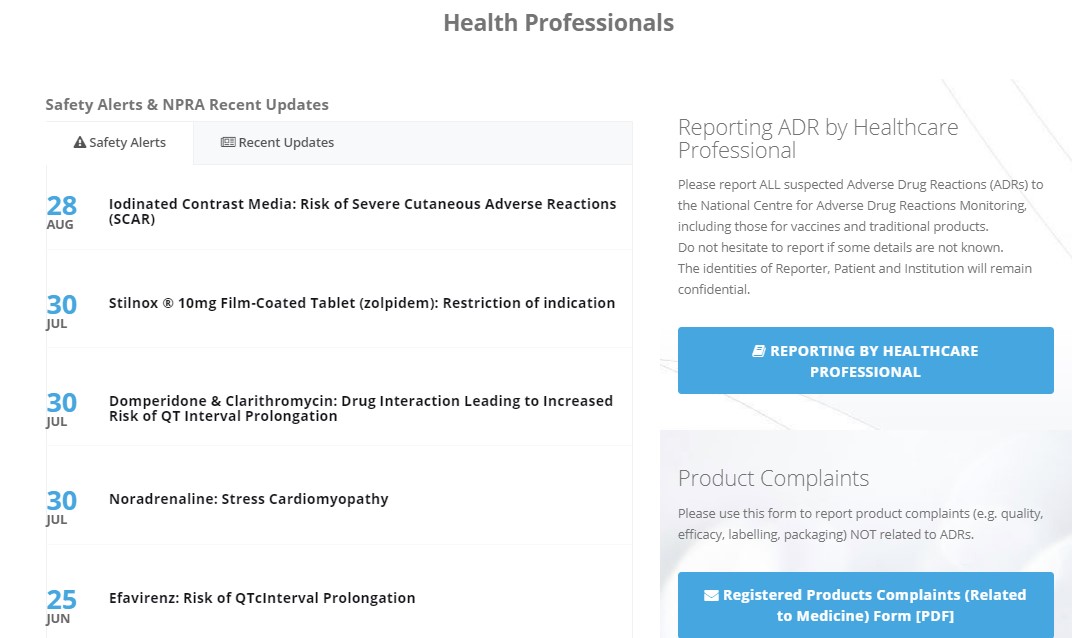
3. Easy to access important information for health professionals at main page
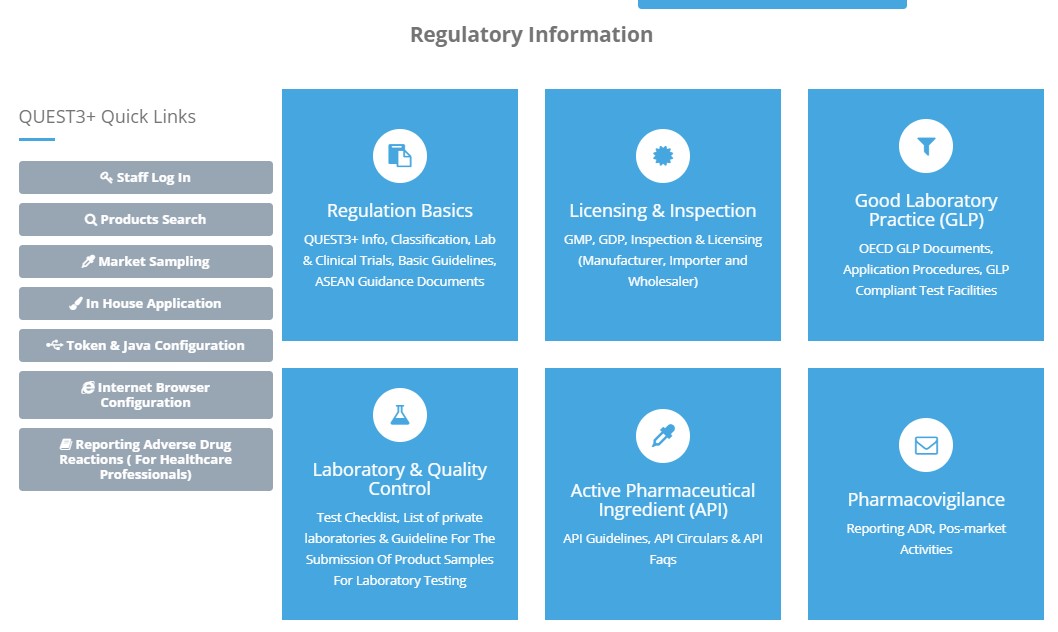
4. User may access guidelines, directives, application forms & recent updates according to regulatory information & product category
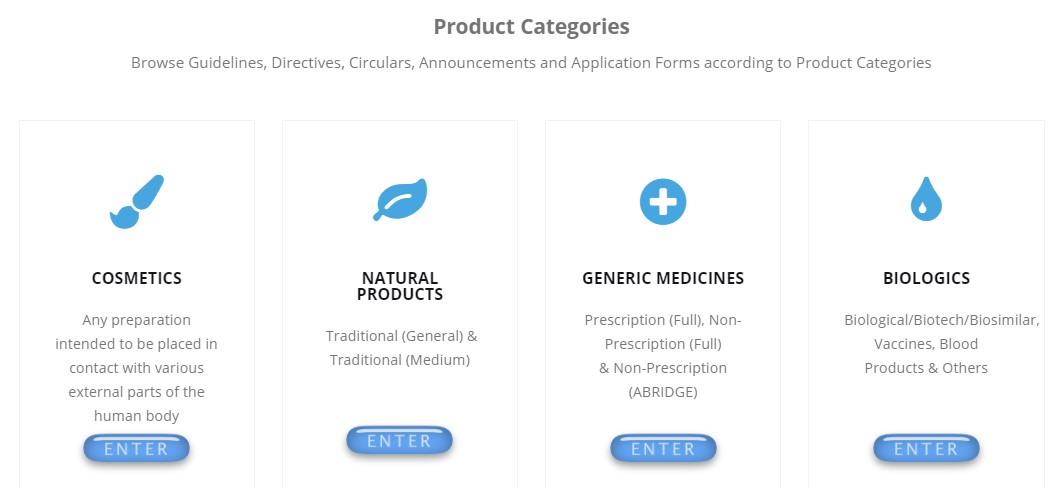
5. Step by step guide for product registration process (currently available for generic medicine only. This product registration process guide will available for all product categories by end of this years)
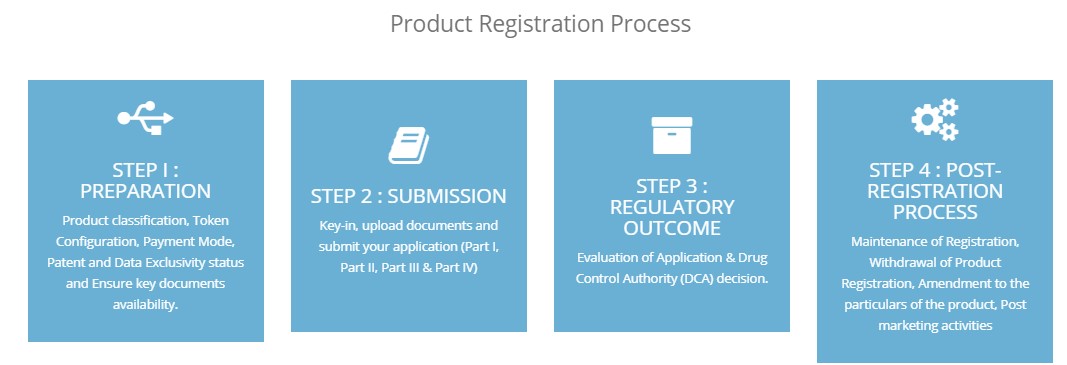
6. Product submission guide according to Part & fields in QUEST3+ system (currently available for generic medicine only. This submission guide will available for all product categories by end of this years)

We're really proud of the new website and feel it will create the experience you're looking for when you pay us a visit.










