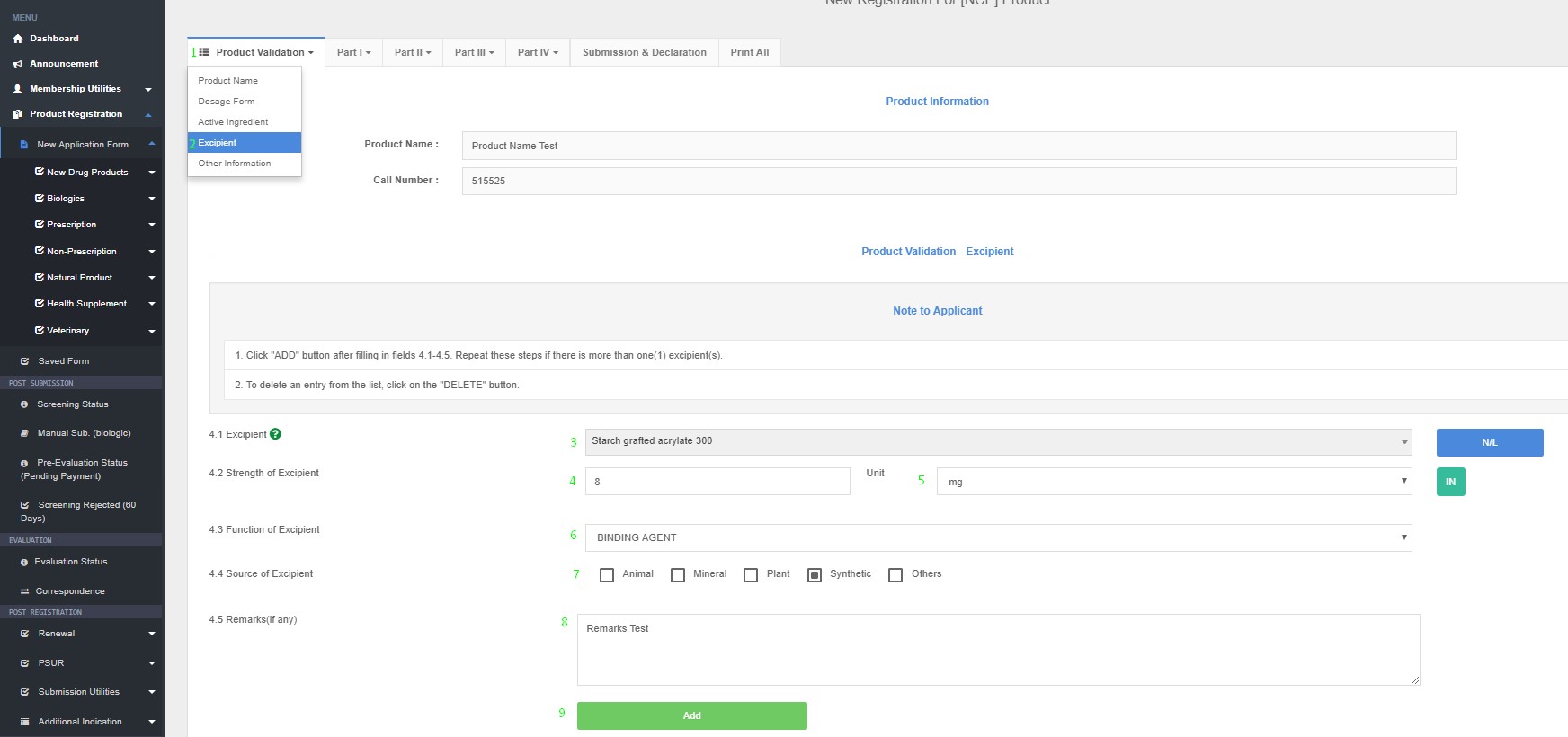
· Please refer Appendix 8.2 List of Prohibited and Restricted Excipients; and Appendix 8.3 Lists of Permitted and Restricted Colouring Agents
- For each excipient ingredient included in a formulation must have a justifiable excipient role and shall be controlled by specifications.
- New excipient will require safety and/or other additional data to support the function in the product prior to addition into the Quest 3 database.
- Please enter function of excipients, e.g. sweetener, preservative, thickening agent, etc. which can be selected from the drop-down list.
How to Access Excipient in QUEST3+ System ?
Product Registration >> New Application Form >> Product Validation >> Excipient