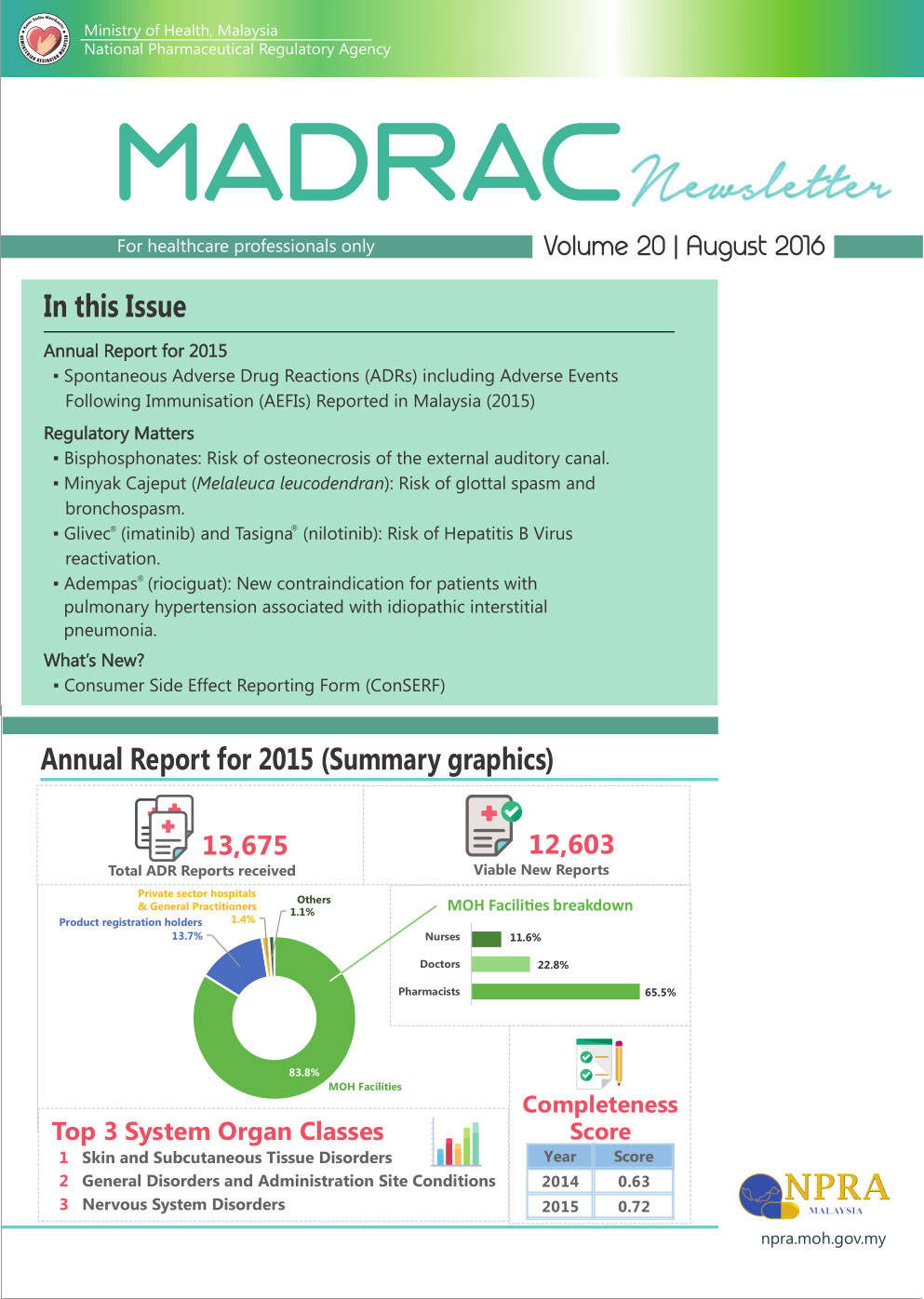
MADRAC Bulletin - August 2016Annual Report for 2015▪ Spontaneous Adverse Drug Reactions (ADRs) including Adverse Events Following Immunisation (AEFIs) Reported in Malaysia Regulatory Matters ▪ Bisphosphonates: Risk of osteonecrosis of the external auditory canal. ▪ Minyak Cajeput (Melaleuca leucodendran): Risk of glottal spasm and bronchospasm. ▪ Glivec (imatinib) and Tasigna (nilotinib): Risk of Hepatitis B Virus reactivation. ▪ Adempas (riociguat): New contraindication for patients with pulmonary hypertension associated with idiopathic interstitial pneumonia. What’s New? ▪ Consumer Side Effect Reporting Form (ConSERF)
|
 DOWNLOAD PDF |









